Main Page
|
Welcome to CannaQAwiki, an encyclopedia for Cannabis and hemp, and the scientific and regulatory aspects that surround them.
Welcome to CannaQAWiki. This wiki continues to be a work in progress, but new content is slowly being added. For example, check out the latest third edition of our guide Past, Present, and Future of Cannabis Laboratory Testing and Regulation in the United States. Additionally, the guide Starting a Cannabis Testing Laboratory has been updated for 2021, and the LIMSpec for Cannabis Testing has been released. Check back soon for additional content!
|
|
Get started
New to CannaQAwiki? We're kind of new to this as well. In case you don't know where to begin, here are some starting points for you: |
Featured article of the month: March
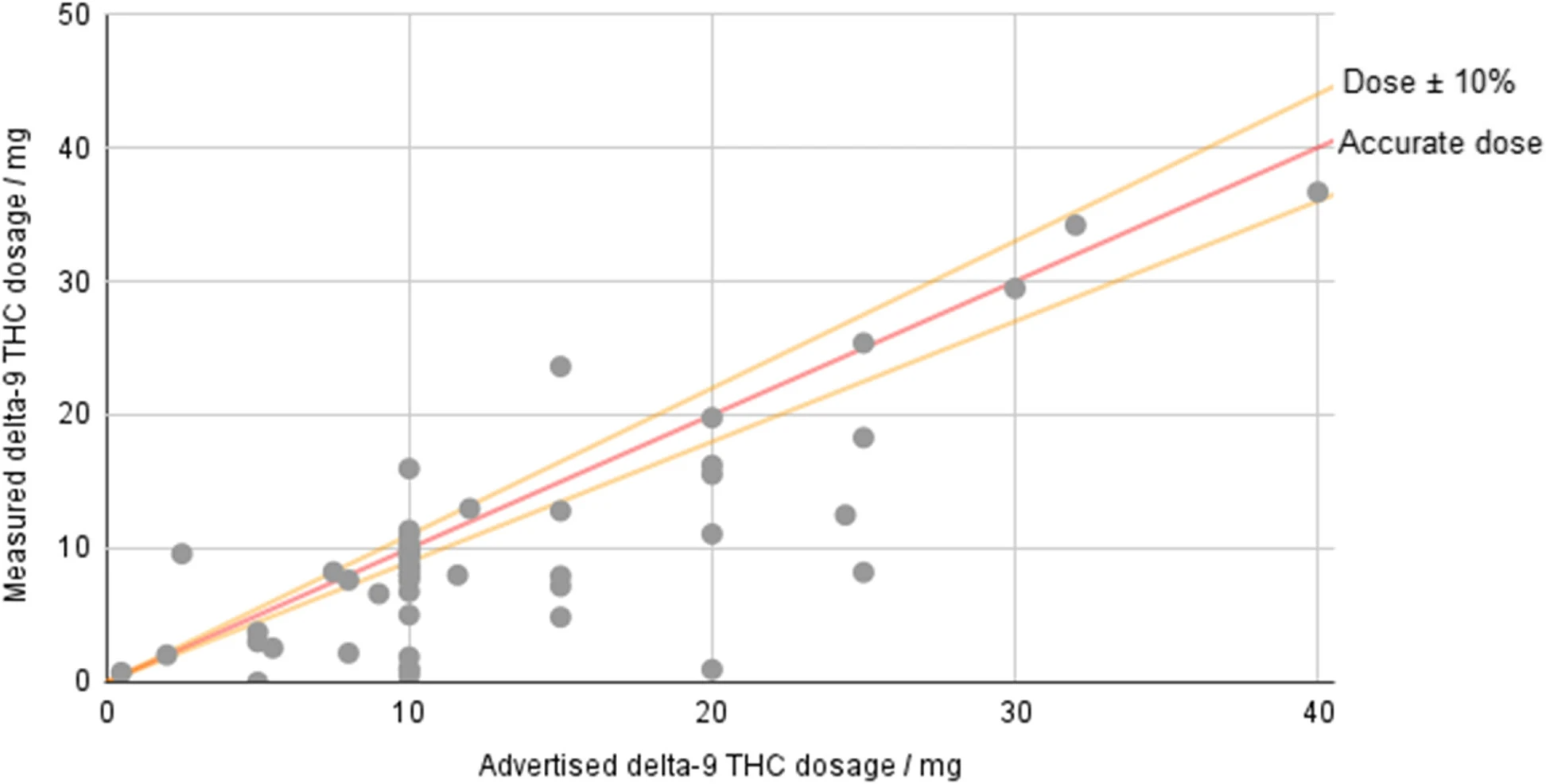
Hemp-derived delta-9-tetrahydrocannabinol (Δ9-THC) products are freely available for sale across much of the USA, but the federal legislation allowing their sale places only minimal requirements on companies. Products must contain no more than 0.3% Δ9-THC by dry weight, but no limit is placed on overall dosage, and there is no requirement that products derived from hemp-based Δ9-THC be tested. However, some states—such as Colorado—specifically prohibit products created by “chemically modifying” a natural hemp component ... (Full article...) Recently featured:
|
|
Articles and reference works
Our wiki is young and will take time to grow. However, here are some of our important topical articles and guides, as well as reference works like lists and tables (which specifically appear in the "RefWork" namespace): Topical articles and guides:
|
CannaQAwiki journal articles
A CannaQAwiki journal article is a selected open-access article relating to scientific aspects of the Cannabis plant, free to republish elsewhere with proper citation. You can browse the collection using the tools below.
|
|
News and updates
June 23, 2023:A third edition of Starting a Cannabis Testing Laboratory has been released. This latest iteration updates statistics, adds a few new regulation updates, and significantly updates many of the guide's references and facts. The update also adds additional information concerning analytical methods, as well as laboratory systems and their role in the cannabis testing lab. Resources were also updated and expanded. Additionally the LIMSpec for Cannabis Testing has been updated to included the latest version of LIMSpec, modified for the cannabis laboratory seeking a laboratory informatics solution!Shawn Douglas (talk) 19:05, 23 June 2023 (UTC)
August 10, 2022:A fourth edition of Past, Present, and Future of Cannabis Laboratory Testing and Regulation in the United States has been released! This latest iteration updates many statistics, adds new regulation updates, and significantly updates many of the guide's resources such as the list of approved cannabis cultivators and testing laboratories. The update also adds additional context to standardization and accreditation efforts, including enforcement and quality subsections. It also adds a few more contextual bits of information about laboratory automation and its role in the cannabis testing lab.Shawn Douglas (talk) 14:31, 10 August 2022 (UTC)
|