Difference between revisions of "Journal:Recent advances in electrochemical sensor technologies for THC detection—A narrative review"
Shawndouglas (talk | contribs) (Saving and adding more.) |
Shawndouglas (talk | contribs) (Saving and adding more.) |
||
| Line 88: | Line 88: | ||
{| border="0" cellpadding="5" cellspacing="0" width="900px" | {| border="0" cellpadding="5" cellspacing="0" width="900px" | ||
|- | |- | ||
| style="background-color:white; padding-left:10px; padding-right:10px;" |<blockquote>'''Fig. | | style="background-color:white; padding-left:10px; padding-right:10px;" |<blockquote>'''Fig. 3''' ('''a''') Optical image of a carbon bead-based sensor. ('''b''') SEM (scanning electron microscope) images of a carbon bead-based sensor. ('''c''') Imaging of a CNT- (carbon nanotube)-based sensor. ('''d''') TEM (transmission electron microscope) image of a carbon bead-based sensor. All imagery taken from Zhang ''et al.'' 2019 (2019) with permission from Elsevier.</blockquote> | ||
|- | |- | ||
|} | |} | ||
| Line 96: | Line 96: | ||
==Sensors developed for THC detection in oral fluid specimens== | ==Sensors developed for THC detection in oral fluid specimens== | ||
Oral fluid or saliva analysis for roadside drug testing is a great substitute for routine blood or urine tests due to being particularly non-invasive while also removing the need for inconvenient observations during sample collection for urine analysis. These tests are normally performed as a screening step. Goodwin ''et al.'' reported on a sensor developed by modification of micro-sized graphite powder by abrasive immobilization of 4-amino-2,6-diphenylphenol onto a basal plane pyrolytic graphite electrode. This sensor was then used for indirect detection of THC in oral fluid. The mechanism under which this sensor operates was described such that by addition of THC, the reduction wave—which corresponds to the electrochemical reduction of quinoneimine (QI) back to aminophenol (AP)—reduces in magnitude due to the reaction between QI and THC, and this, in turn, provides a useful analytical signal. This technique was indicated as having potential because it does not involve direct oxidation of THC, which can cause electrode passivation. Goodwin ''et al.'' also reported that such a device has the potential to detect THC using screen-printed electrodes and similar approaches. The construction of the sensor was done through machining pyrolytic graphite discs into a 4.9 mm diameter. The counter electrode was a platinum wire, and the measurements were performed against a saturated calomel electrode. The linear dynamic range of the sensor was reported to be from 1.25 to 25 μM, with a limit of detection of 1 μM. (Goodwin et al. 2006) | |||
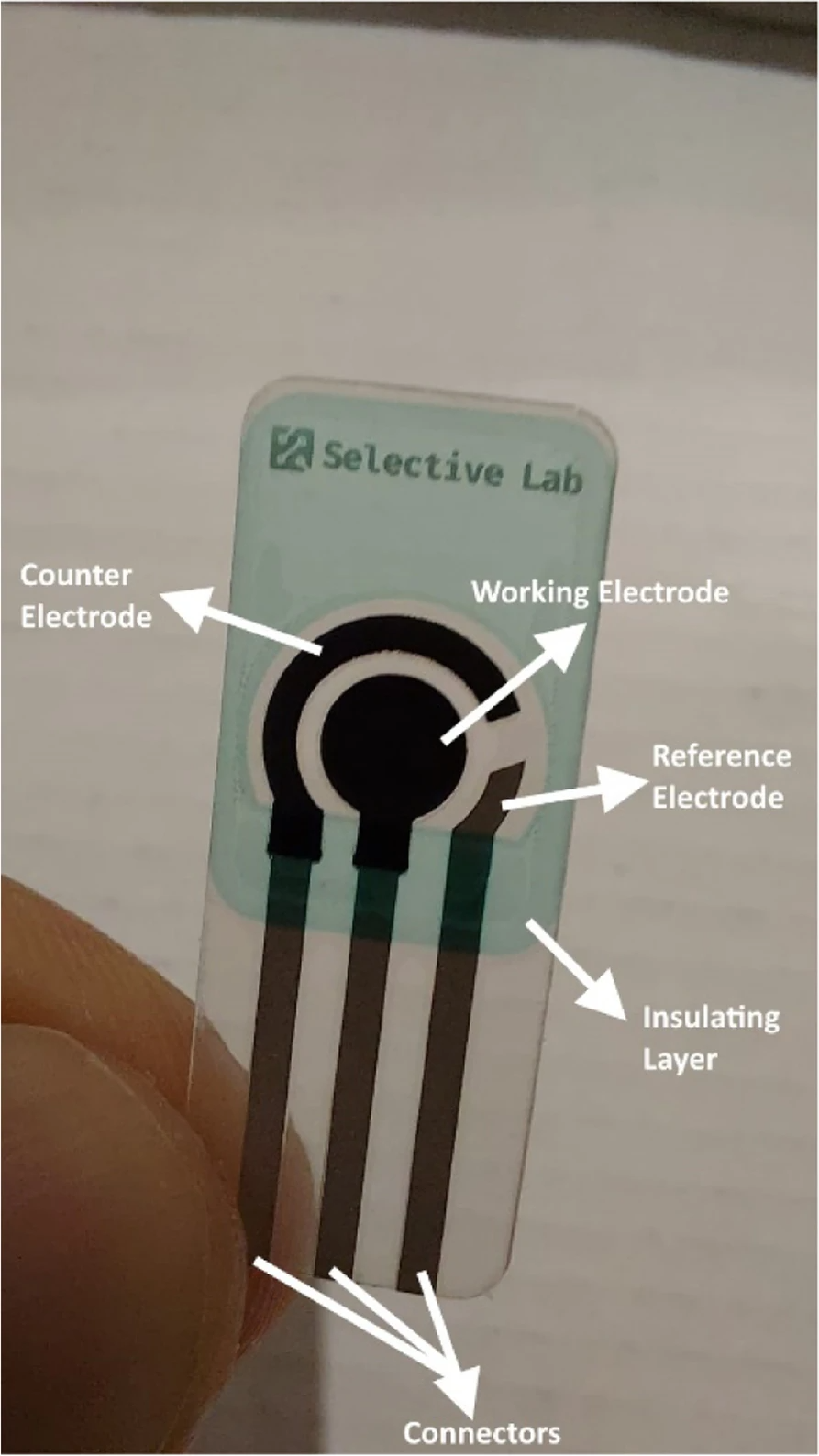
Modern screen-printed electrodes are produced by using different kinds of inks on various types of plastic or ceramic substrates. A wide variety of these screen-printed electrodes are commercially available. The versatility of screen-printed electrodes is found in the wide range of possibilities for the modification of the electrodes. The composition of the inks can be modified by addition of different substances such as metals, enzymes, polymers, complexing agents, etc. It is also possible to modify the electrodes by depositing different substances on the surface of the electrodes such as metal films, polymers, enzymes, etc. (Suresh et al. 2021) Figure 4 illustrates a screen-printed electrode and its different parts. | |||
[[File:Fig4 Amini JofCannRes22 4.png|400px]] | |||
{{clear}} | |||
{| | |||
| style="vertical-align:top;" | | |||
{| border="0" cellpadding="5" cellspacing="0" width="400px" | |||
|- | |||
| style="background-color:white; padding-left:10px; padding-right:10px;" |<blockquote>'''Fig. 4''' A screen-printed electrode and its different parts</blockquote> | |||
|- | |||
|} | |||
|} | |||
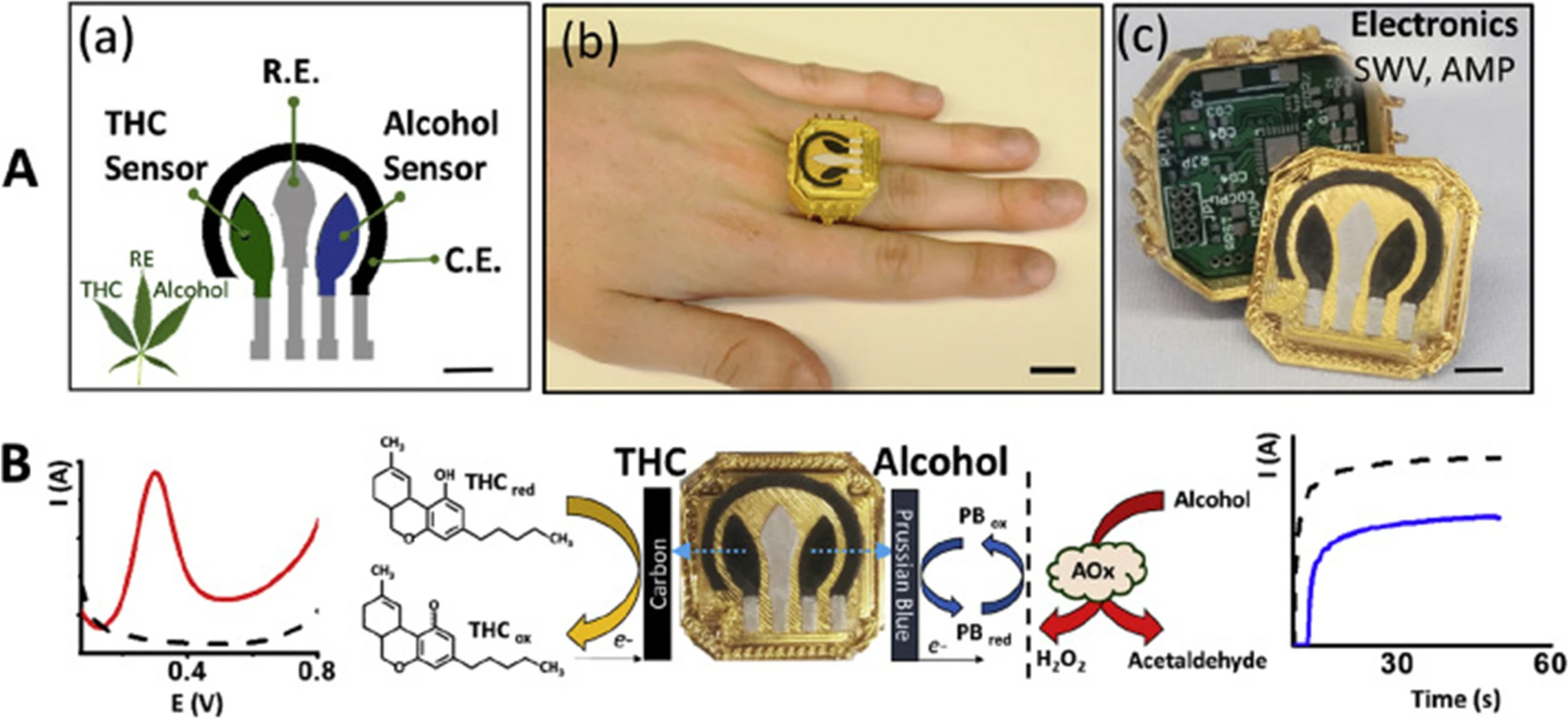
In another interesting report, Mishra ''et al.'' (Mishra et al. 2020) presented work on a wearable electrochemical sensor for the simultaneous direct, decentralized detection of THC and alcohol in oral fluids. This sensor was designed in the form of a ring and includes a square-wave voltammetric THC sensor and an amperometric enzymatic alcohol sensor on the ring cap, with the wireless electronics embedded within the ring case. The disposable sensing electrode ring cap was designed so that it allows for fast replacement through alignment with the spring-loaded pins, mounted on the printed circuit board (PCB), with the current collectors of the sensing electrodes, after each oral fluids test. The printed sensor for dual-analyte detection (ring cover) was composed of a multi-wall carbon nanotube/carbon electrode for THC detection and a Prussian-blue transducer, coated with an alcohol oxidase/chitosan reagent layer, for alcohol detection. This structure makes it possible for THC and alcohol to be detected simultaneously in the same diluted oral fluids specimen in three minutes without any interference from the matrix and also no cross-talk among sensors. Figure 5 illustrates the ring sensor developed by Mishra ''et al.'' The detection limits were indicted to be 0.5 μM for THC and 0.2 mM for alcohol. This wearable THC/alcohol sensor was found to be promising for both roadside drug testing and for a drivers’ self-assessment before driving. (Mishra et al. 2020) | |||
[[File:Fig5 Amini JofCannRes22 4.png|900px]] | |||
{{clear}} | |||
{| | |||
| style="vertical-align:top;" | | |||
{| border="0" cellpadding="5" cellspacing="0" width="900px" | |||
|- | |||
| style="background-color:white; padding-left:10px; padding-right:10px;" |<blockquote>'''Fig. 5''' ('''A''') (a) The sensor designed by Mishra ''et al.'' for simultaneous detection of THC and alcohol. (b) Image of their ring-shaped sensor. (c) The ring polymeric case, with the embedded electronics and replaceable screen-printed electrodes shown. ('''B''') Mechanisms of THC and alcohol detection using the ring sensor. The voltammogram for the detection of THC (in red) and the amperogram of alcohol detection is also illustrated (in blue). Taken from Mishra ''et al.'' 2020 (2020) with permission from Elsevier</blockquote> | |||
|- | |||
|} | |||
|} | |||
In another work, Nissim and Compton (Nissim and Compton 2015) introduced an optimized carbon paste electrode, made from graphite powder and mineral oil, for sensitive detection of THC in both aqueous solutions of pH 10.0 and in synthetic oral fluids. Absorptive stripping voltammetry was utilized as the detection technique. A copper rod with a radius of 1.97 mm running through a Teflon rod was used to fabricate the carbon paste electrode. The copper rod embedded in the Teflon rod was adjusted so that it left a 1.00 mm-deep cavity at the edge. Two sorts of pastes were used, which were made by mixing graphite powder with either dioctyl phthalate or mineral oil. 1.4 mL dioctyl phthalate and 4.26 g graphite powder were mixed to prepare the graphite/dioctyl phthalate paste. The same ratio also was used to prepare the graphite/mineral oil paste. The surface of the carbon paste electrode was renewed between each scan by packing it with fresh paste. Practical limits of detection for THC using this sensor were reported to be 0.50 μM and 0.10 μM in stationary and stirred aqueous borate buffer solutions, respectively, whereas the theoretical limits of detections were calculated to be 0.48 nM and 0.41 nM for stationary and stirred THC aqueous borate buffer solutions, respectively. This sensor was capable of detecting THC concentrations as low as 0.50 μM in synthetic oral fluids solutions. The sensor had sensitivities of 0.12 μA μM−1, 0.84 μA μM−1, and 0.067 μA μM−1 for the stationary buffer, the stirred buffer, and the oral fluids matrix, respectively. (Nissim and Compton 2015) | |||
Revision as of 18:26, 29 May 2022
| Full article title | Recent advances in electrochemical sensor technologies for THC detection—A narrative review |
|---|---|
| Journal | Journal of Cannabis Research |
| Author(s) | Amini, Kaveh; Sepehrifard, Ali; Valinasabpouri, Ali; Safruk, Jennifer; Angelone, Davide; de Campos Lourenco, Tiago |
| Author affiliation(s) | Selective Lab, Inc. |
| Primary contact | Email: kamini at selectivelab dot com |
| Year published | 2022 |
| Volume and issue | 4 |
| Article # | 12 |
| DOI | 10.1186/s42238-022-00122-3 |
| ISSN | 2522-5782 |
| Distribution license | Creative Commons Attribution 4.0 International |
| Website | https://jcannabisresearch.biomedcentral.com/articles/10.1186/s42238-022-00122-3 |
| Download | https://jcannabisresearch.biomedcentral.com/track/pdf/10.1186/s42238-022-00122-3.pdf (PDF) |
|
|
This article should be considered a work in progress and incomplete. Consider this article incomplete until this notice is removed. |
Abstract
Background: delta-9-tetrahydrocannabinol (Δ9-THC or simply THC) is the main psychoactive component and one of the most important medicinal compounds in cannabis. Whether in human body fluids and breath or in laboratory and field samples, rapid and easy detection of THC is crucial. It provides insights into the impact of THC on the human organism, as well as its medicinal benefits. It also guides cannabis growers in determining different stages of the growth of the plant in the field, and eventually it helps scientists in the laboratory to assure the quality of the products and determine their potency or better understand product development procedures. The significance of fast THC detection in forensic analysis also cannot be overlooked. Electrochemical sensor technologies show promise in fulfilling the need for fast, easy, and low-cost detection of THC.
Method: In this work, we review the recent advances in sensor technologies developed for the purpose of fast and accurate THC detection. Research performed mostly in the past decade, and that detecting THC directly without any derivatization, was the main target of this review. The scope of this narrative review was reports on detecting THC in synthetic samples and plants as well as oral fluid.
Results: Electrochemical sensor technologies are sensitive enough and have the potential for fast, easy, and low-cost detection of THC for roadside testing, THC trending in growing cannabis plants, THC product development, and formulation for medical purposes, etc. They can also provide an alternative for costly chromatography and mass spectrometry-based methods.
Conclusion: The main challenges facing electrochemical sensors, however, are nonspecific interaction and the interference of compounds and species from the matrix. Special requirement for storing sensors modified with antibodies or proteins is another challenge in this field. Preparing long-lasting and reusable sensors is a field worthy of attention.
Keywords: Δ9-tetrahydrocannabinol, THC, sensor, cannabis, electrochemical detection
Introduction
In light of the legalization of medicinal and recreational cannabis in Canada and many states in the United Stats, as well as other countries around the world, a great deal of attention has been given to the research in different areas of cannabis chemistry. (Crean et al. 2011) Additionally, a growing body of evidence for the therapeutic effects of cannabis has turned cannabis research into a hot topic. (Hill 2015; Hoffmann and Weber 2010) Over 60 unique cannabinoids have been identified in the Cannabis plant. (Vemuri and Makriyannis 2015) Out of these cannabinoids, delta-9-tetrahydrocannabinol (Δ9-THC or simply THC), cannabidiol (CBD), and cannabinol (CBN) are the most significant ones. (Grotenhermen 2003) THC is responsible for the psychoactive property of cannabis and causes euphoria, drowsiness, hallucinations, and temporal distortions. (Ashton 2001) CBD, another significant cannabinoid, on the other hand is not psychoactive; however, it has neuroprotective, sedating, anti-inflammatory, and analgesic impacts. (Chakravarti et al. 2014; Hill 2019; Klimuntowski et al. 2020; Mechoulam et al. 2007)
However, the legalization of cannabis has also raised concerns about driving under the influence of THC. An increase in the number of cases of THC-impaired driving has been reported in regions of the world where cannabis has been legalized. (Kalant 2001; Zuardi 2006; Kim et al. 2013) This has led to questions and doubts about how to best approach testing for potential THC levels in drivers.[1] Electrochemical sensors may make for one possible approach to this problem, as well for other cannabis testing applications.
In this work, we critically review the recent developments and advances in sensor technologies THC detection, as well as the potential presented for different applications, from forensic and law enforcement to quality control and product development. First, a brief background on electrochemical sensors and their potential in THC analysis is given. Then sensors will be discussed in two major categories: (a) early applications in synthetic solutions and plant analysis and (b) applications in oral fluid analysis. Sensors focused on THC detection in breath, which are thoroughly reviewed by Ramzy and Priefer (Ramzy and Priefer 2021), have not been included in this review.
Background on electrochemical sensors for THC analysis
Electrochemical sensors provide a highly sensitive tool for the analysis of THC, and they also have advantages such as easy miniaturization and usability in turbid matrices. In these sensors, the current or potential change, which occurs as a result of the interactions or reactions at the interface between the sensor surface and the samples solution, is measured. On the basis of the electrochemical technique used for detection, these sensors can be broadly categorized as potentiometric, impedimetric, voltammetric, and amperometric.[2] In potentiometric sensors, measurements are based on the development of electrochemical potential in proportion to the activity of the analyte.[3] In impedimetric sensors, electrochemical impedance spectroscopy (EIS) is employed as the detection technique. In these sensors, a low voltage sinusoidal potential is applied at different frequencies to the sensor and the impedance is measured as a function of frequencies using the resulting current. The interaction between the analyte and a biorecognition element immobilized on the sensor surface will cause changes in the impedance. The results will be interpreted in terms of an equivalent circuit.[4] (Another major advantage of impedance-based sensors is being label-free.[5]) Voltammetric sensors employ electrochemical techniques such as cyclic voltammetry, square wave voltammetry, and differential pulse voltammetry, whereas in the case of amperometric sensors, the changes in current are followed.[6]
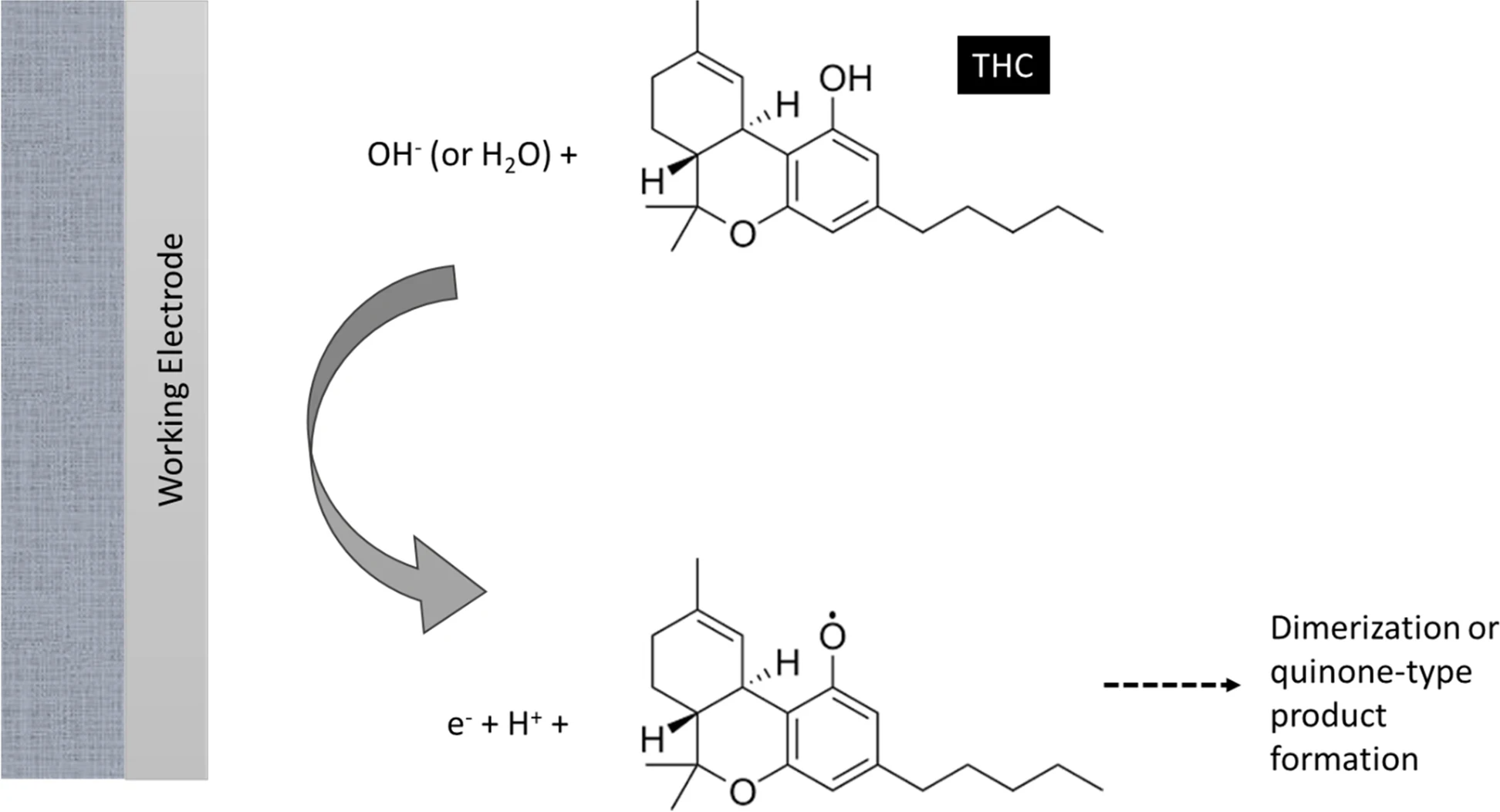
Figure 1 illustrates the mechanism under which electrochemical sensors operate towards the detection of THC, according to Renaud-Young et al. (2019)
|
The standard THC detection method includes costly, complex, and time-consuming steps of obtaining a clinical specimen or botanical sample and analyzing it using chromatography with mass spectrometry detection. This method obviously would be cumbersome when it comes to roadside testing or testing plant material directly in the field. The limitations of standard THC detection methods for these testing purposes have motivated scientists to develop portable, non-invasive, fast, and low-cost sensor technologies for on-site THC screening and testing. (Sivashanmugan et al. 2019) Sensors capable of detecting and quantifying THC and other cannabinoids can also be very effective in other areas such as monitoring different stages of plant growth, performing quality control on cannabis products, and while developing products from cannabis. Also, ease-of-use—alongside rapid detection—for these sensors makes them ideal to be used by dispensaries or consumers to evaluate the THC levels or THC-CBD ratio of the products they acquire. (Comeau et al. 2019) In comparison to other portable methods, sensor-based technologies have the potential to reduce analysis time and cost, as well as improve sensitivity through the process of miniaturization (Amini and Kraatz 2015a, b; Amini and Kraatz 2016; Jadon et al. 2016), offering a novel and alternative approach to testing cannabinoids like THC for different purposes.
Development of early sensors and trials in synthetic solutions and plant extracts
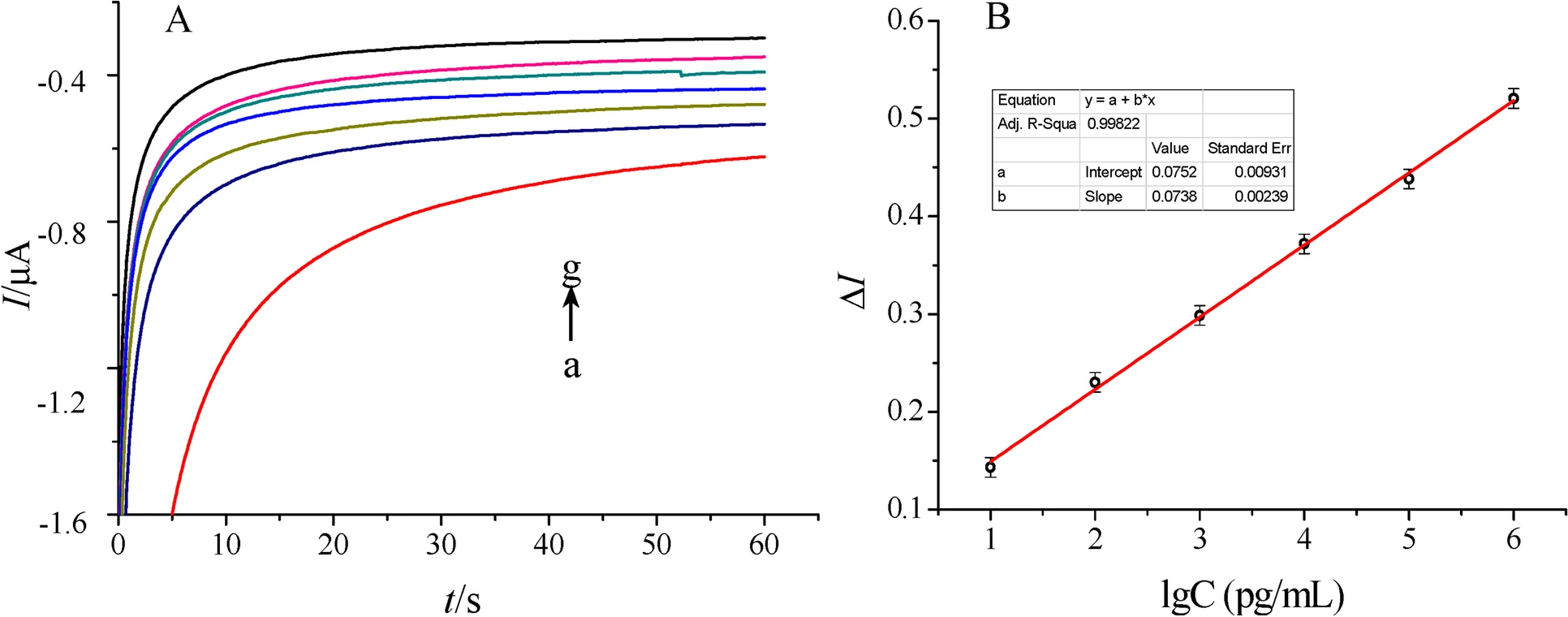
The first attempt towards the development of a THC sensor has been reported by Dingqiang et al. (2016) To develop this sensor, a THC antibody derived from Balb/c mice as the bio-recognition element was immobilized on a novel double-layer gold nanoparticle electrode. Also, an electrochemical biosensing signal amplification system with gold nanoparticle-thionine-chitosan absorbing horseradish peroxidase (HRP) was used to enhance the number of immobilized antibodies and thus the electrochemical signal. Developed biosensors were employed to determine THC in phosphate buffer saline (PBS) using the amperometric I-t curve method. The dynamic linear range of the calibration curve made by plotting response current versus THC concentration was from 0.01~103 ng/mL, with a correlation coefficient of 0.9986. The lowest limit of detection for THC also was reported to be 3.3 pg/mL (S/N = 3), with good sensitivity and reproducibility. Current response curves for determination of THC and the calibration curve constructed in the work of Dingqiang et al. (2016) are illustrated in Fig. 2.
|
More recently, Zhang et al. employed carbon nanotubes (CNT) or carbon beads and poly(methyl acrylic acid-co-ethylene glycol dimethacrylate) (poly(MAA-Co-EGDMA)) with molecularly imprinting technology in micropipette tubes to develop THC sensors. (Zhang et al. 2019) Molecularly imprinted polymers (MIPs) are synthetic receptors which can selectively bind to their target molecules and, therefore, can be used as recognition elements in sensors as a replacement for relatively unstable bio-recognition elements such as enzymes and antibodies. Zhang et al. emphasized the importance of utilizing carbon materials in sensors, which results in a high sensitivity because of the high surface area, as well as appropriate MIPs technique to create THC molecular cavities on the surface of carbon micro-beads and carbon nanotubes (CNT) packed in plastic micropipette tips. These THC MIPs were synthesized by copolymerization of methacrylic acid (MAA) and ethylene glycol dimethacrylate (EDGMA) in presence of THC initiated with 4,4′-azobis (4-cyanovaleric Acid) (AIBN), and then THC was removed to generate THC molecular cavities for specific binding of THC. Optical and microscopic images of their developed sensors are shown in Fig. 3. The developed sensors showed high selectivity towards THC over caffeine and acetaminophen. The detection limit of THC using CNT-MIP sensors was determined to be 0.18 ± 0.02 ng/mL, which is significantly improved in comparison to the detection limit using nonimprinted polymers (NIP), which was 12.5 ± 0.5 ng/mL. (Zhang et al. 2019)
|
Another work employing MIPs was introduced by Canfarotta et al. (Canfarotta et al. 2018) In their research, a manufacturing-friendly protocol for integration of MIP nanoparticles (nanoMIPs) with a (label-free) capacitive sensor was developed. The two templates for which the nanoMIPs were produced included THC as a small molecule and trypsin as a protein. Using this sensor, determining THC was demonstrated to be possible at physiological concentrations. Another advantage of these sensors was the possibility of using them for detection and quantification of other biomolecules by varying the nanoMIPs. These nanoMIPs can then be virtually produced against any target. (Canfarotta et al. 2018)
Sensors developed for THC detection in oral fluid specimens
Oral fluid or saliva analysis for roadside drug testing is a great substitute for routine blood or urine tests due to being particularly non-invasive while also removing the need for inconvenient observations during sample collection for urine analysis. These tests are normally performed as a screening step. Goodwin et al. reported on a sensor developed by modification of micro-sized graphite powder by abrasive immobilization of 4-amino-2,6-diphenylphenol onto a basal plane pyrolytic graphite electrode. This sensor was then used for indirect detection of THC in oral fluid. The mechanism under which this sensor operates was described such that by addition of THC, the reduction wave—which corresponds to the electrochemical reduction of quinoneimine (QI) back to aminophenol (AP)—reduces in magnitude due to the reaction between QI and THC, and this, in turn, provides a useful analytical signal. This technique was indicated as having potential because it does not involve direct oxidation of THC, which can cause electrode passivation. Goodwin et al. also reported that such a device has the potential to detect THC using screen-printed electrodes and similar approaches. The construction of the sensor was done through machining pyrolytic graphite discs into a 4.9 mm diameter. The counter electrode was a platinum wire, and the measurements were performed against a saturated calomel electrode. The linear dynamic range of the sensor was reported to be from 1.25 to 25 μM, with a limit of detection of 1 μM. (Goodwin et al. 2006)
Modern screen-printed electrodes are produced by using different kinds of inks on various types of plastic or ceramic substrates. A wide variety of these screen-printed electrodes are commercially available. The versatility of screen-printed electrodes is found in the wide range of possibilities for the modification of the electrodes. The composition of the inks can be modified by addition of different substances such as metals, enzymes, polymers, complexing agents, etc. It is also possible to modify the electrodes by depositing different substances on the surface of the electrodes such as metal films, polymers, enzymes, etc. (Suresh et al. 2021) Figure 4 illustrates a screen-printed electrode and its different parts.
|
In another interesting report, Mishra et al. (Mishra et al. 2020) presented work on a wearable electrochemical sensor for the simultaneous direct, decentralized detection of THC and alcohol in oral fluids. This sensor was designed in the form of a ring and includes a square-wave voltammetric THC sensor and an amperometric enzymatic alcohol sensor on the ring cap, with the wireless electronics embedded within the ring case. The disposable sensing electrode ring cap was designed so that it allows for fast replacement through alignment with the spring-loaded pins, mounted on the printed circuit board (PCB), with the current collectors of the sensing electrodes, after each oral fluids test. The printed sensor for dual-analyte detection (ring cover) was composed of a multi-wall carbon nanotube/carbon electrode for THC detection and a Prussian-blue transducer, coated with an alcohol oxidase/chitosan reagent layer, for alcohol detection. This structure makes it possible for THC and alcohol to be detected simultaneously in the same diluted oral fluids specimen in three minutes without any interference from the matrix and also no cross-talk among sensors. Figure 5 illustrates the ring sensor developed by Mishra et al. The detection limits were indicted to be 0.5 μM for THC and 0.2 mM for alcohol. This wearable THC/alcohol sensor was found to be promising for both roadside drug testing and for a drivers’ self-assessment before driving. (Mishra et al. 2020)
|
In another work, Nissim and Compton (Nissim and Compton 2015) introduced an optimized carbon paste electrode, made from graphite powder and mineral oil, for sensitive detection of THC in both aqueous solutions of pH 10.0 and in synthetic oral fluids. Absorptive stripping voltammetry was utilized as the detection technique. A copper rod with a radius of 1.97 mm running through a Teflon rod was used to fabricate the carbon paste electrode. The copper rod embedded in the Teflon rod was adjusted so that it left a 1.00 mm-deep cavity at the edge. Two sorts of pastes were used, which were made by mixing graphite powder with either dioctyl phthalate or mineral oil. 1.4 mL dioctyl phthalate and 4.26 g graphite powder were mixed to prepare the graphite/dioctyl phthalate paste. The same ratio also was used to prepare the graphite/mineral oil paste. The surface of the carbon paste electrode was renewed between each scan by packing it with fresh paste. Practical limits of detection for THC using this sensor were reported to be 0.50 μM and 0.10 μM in stationary and stirred aqueous borate buffer solutions, respectively, whereas the theoretical limits of detections were calculated to be 0.48 nM and 0.41 nM for stationary and stirred THC aqueous borate buffer solutions, respectively. This sensor was capable of detecting THC concentrations as low as 0.50 μM in synthetic oral fluids solutions. The sensor had sensitivities of 0.12 μA μM−1, 0.84 μA μM−1, and 0.067 μA μM−1 for the stationary buffer, the stirred buffer, and the oral fluids matrix, respectively. (Nissim and Compton 2015)
References
- ↑ Pearlson, Godfrey D.; Stevens, Michael C.; D'Souza, Deepak Cyril (24 September 2021). "Cannabis and Driving". Frontiers in Psychiatry 12: 689444. doi:10.3389/fpsyt.2021.689444. ISSN 1664-0640. PMC PMC8499672. PMID 34630173. https://www.frontiersin.org/articles/10.3389/fpsyt.2021.689444/full.
- ↑ Dzulkurnain, Nurul Akmaliah; Mokhtar, Marliyana; Rashid, Jahwarhar Izuan Abdul; Knight, Victor Feizal; Wan Yunus, Wan Md Zin; Ong, Keat Khim; Mohd Kasim, Noor Azilah; Mohd Noor, Siti Aminah (15 August 2021). "A Review on Impedimetric and Voltammetric Analysis Based on Polypyrrole Conducting Polymers for Electrochemical Sensing Applications" (in en). Polymers 13 (16): 2728. doi:10.3390/polym13162728. ISSN 2073-4360. PMC PMC8401594. PMID 34451266. https://www.mdpi.com/2073-4360/13/16/2728.
- ↑ Yunus, Sami; Jonas, Alain M.; Lakard, Boris (2013), Roberts, Gordon C. K., ed., "Potentiometric Biosensors" (in en), Encyclopedia of Biophysics (Berlin, Heidelberg: Springer Berlin Heidelberg): 1941–1946, doi:10.1007/978-3-642-16712-6_714, ISBN 978-3-642-16711-9, http://link.springer.com/10.1007/978-3-642-16712-6_714. Retrieved 2022-05-29
- ↑ Bahadır, Elif Burcu; Sezgintürk, Mustafa Kemal (2 January 2016). "A review on impedimetric biosensors" (in en). Artificial Cells, Nanomedicine, and Biotechnology 44 (1): 248–262. doi:10.3109/21691401.2014.942456. ISSN 2169-1401. https://www.tandfonline.com/doi/full/10.3109/21691401.2014.942456.
- ↑ Daniels, Jonathan S.; Pourmand, Nader (1 June 2007). "Label-Free Impedance Biosensors: Opportunities and Challenges" (in en). Electroanalysis 19 (12): 1239–1257. doi:10.1002/elan.200603855. PMC PMC2174792. PMID 18176631. https://onlinelibrary.wiley.com/doi/10.1002/elan.200603855.
- ↑ Tajik, Somayeh; Dourandish, Zahra; Jahani, Peyman Mohammadzadeh; Sheikhshoaie, Iran; Beitollahi, Hadi; Shahedi Asl, Mehdi; Jang, Ho Won; Shokouhimehr, Mohammadreza (2021). "Recent developments in voltammetric and amperometric sensors for cysteine detection" (in en). RSC Advances 11 (10): 5411–5425. doi:10.1039/D0RA07614G. ISSN 2046-2069. PMC PMC8694840. PMID 35423079. http://xlink.rsc.org/?DOI=D0RA07614G.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation. Some grammar and punctuation was cleaned up to improve readability. In some cases important information was missing from the references, and that information was added. The discussion about driving under the influence of THC was deemed too abrupt, and a few extra sentences and a citation were added to help the introduction flow better into discussion about electrochemical sensors. Additionally, the original has an introduction section that leads directly into a conclusion section, which is awkward; in this version, content has been divided up into more sections to better segue from the introduction to the conclusion. The Amini and Kratz citations of the original do not back up the claims about different electrochemical sensor modes; they were omitted for this version and more relevant citations were added in the discussion of electrochemical sensor modes.